PERSONALIZED BRAIN FUNCTION SECTORS (PBFS) AND THEIR APPLICATION IN THE TREATMENT OF NEURODEGENERATIVE DISEASE
2026-04-29
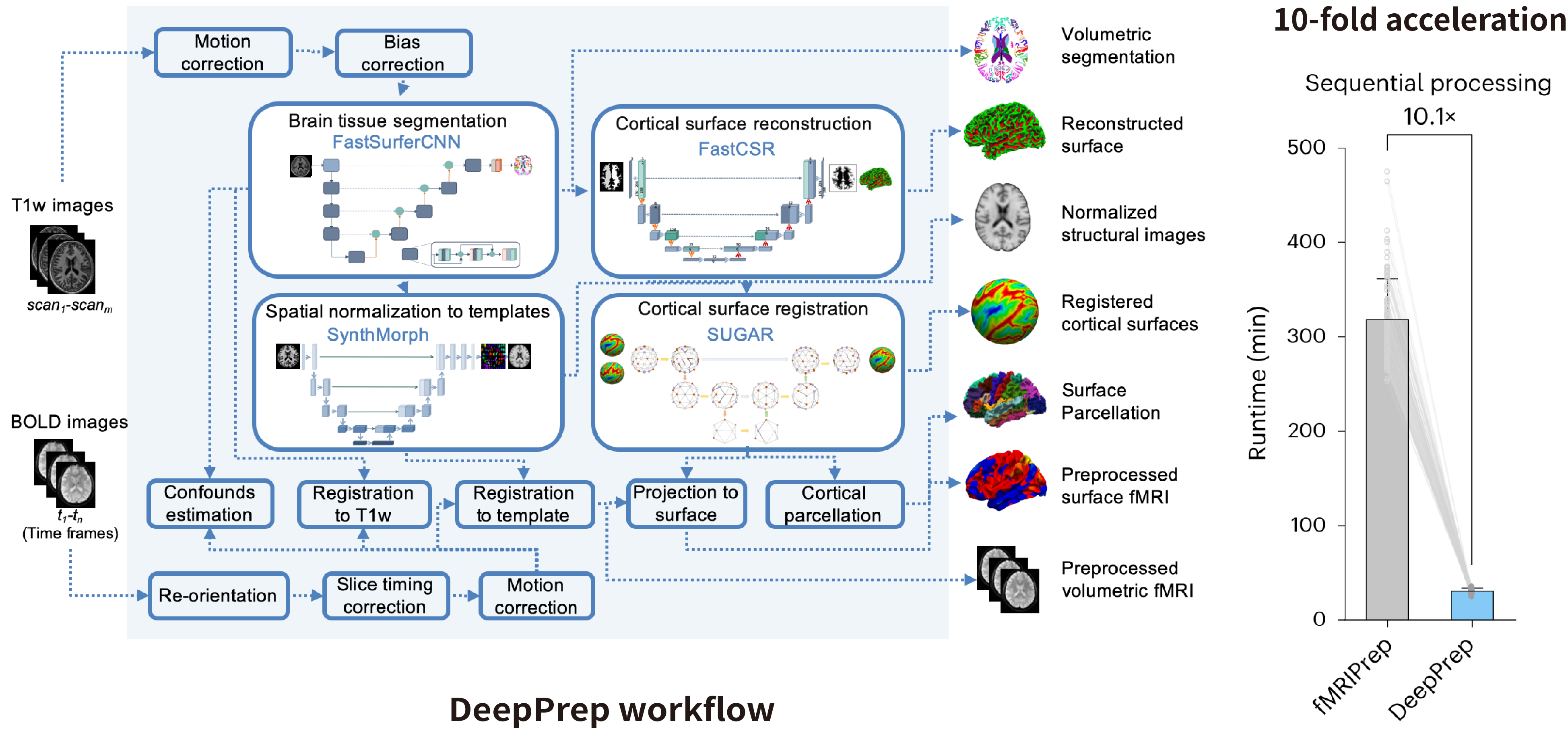
pBFS Foundation: DeepPrep – an accelerated, scalable, and robust deep learning–powered neuroimaging preprocessing pipeline
Neuroimaging is entering the era of big data, driven by large-scale studies, data sharing initiatives, and international consortia. Accurate analyses require complex, multi-stage preprocessing pipelines. Traditional pipelines are often time-consuming and difficult to scale, limiting the ability to leverage massive neuroimaging datasets effectively.
We introduce DeepPrep, a deep learning–enhanced preprocessing pipeline equipped with an advanced workflow manager. Evaluated on over 50,000 scans, DeepPrep achieves a 10-fold acceleration compared to conventional pipelines, while demonstrating exceptional scalability and robustness. By dramatically reducing processing time and ensuring reliable performance across large datasets, DeepPrep addresses a critical bottleneck in neuroimaging and enables efficient, large-scale analyses in the era of big data.
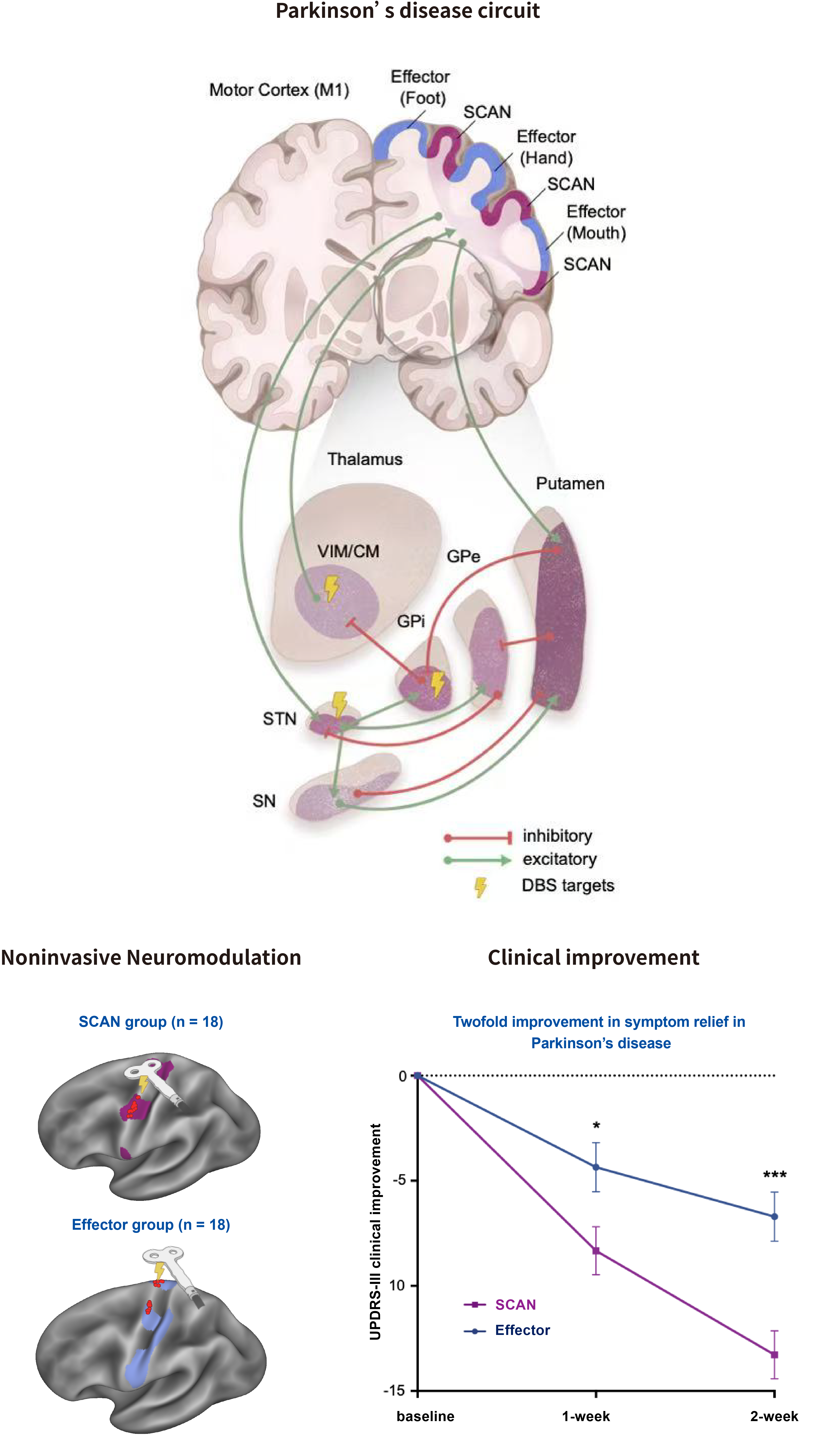
pBFS Treatment: Parkinson’s disease as a somato-cognitive action network disorder
Parkinson’s disease (PD) is a progressive neurological disorder marked by motor and cognitive impairments. Dysfunction of the somato-cognitive action network (SCAN) may underlie PD’s diverse symptoms. We established a large multimodal imaging cohort (n = 863) across multiple interventions, including medication, deep brain stimulation (DBS), transcranial magnetic stimulation (TMS), and MRI-guided focused ultrasound, to investigate SCAN’s role in PD.
Resting-state functional connectivity analyses showed that the substantia nigra and major DBS targets are preferentially connected to SCAN rather than motor areas, with PD characterized by SCAN–subcortical hyperconnectivity. Across interventions, effective treatments consistently reduced this hyperconnectivity. In a randomized trial of 36 patients, repetitive TMS targeting SCAN produced roughly twice the motor improvement compared to motor cortex stimulation, highlighting SCAN hyperconnectivity as a core PD pathology and a superior neuromodulation target.
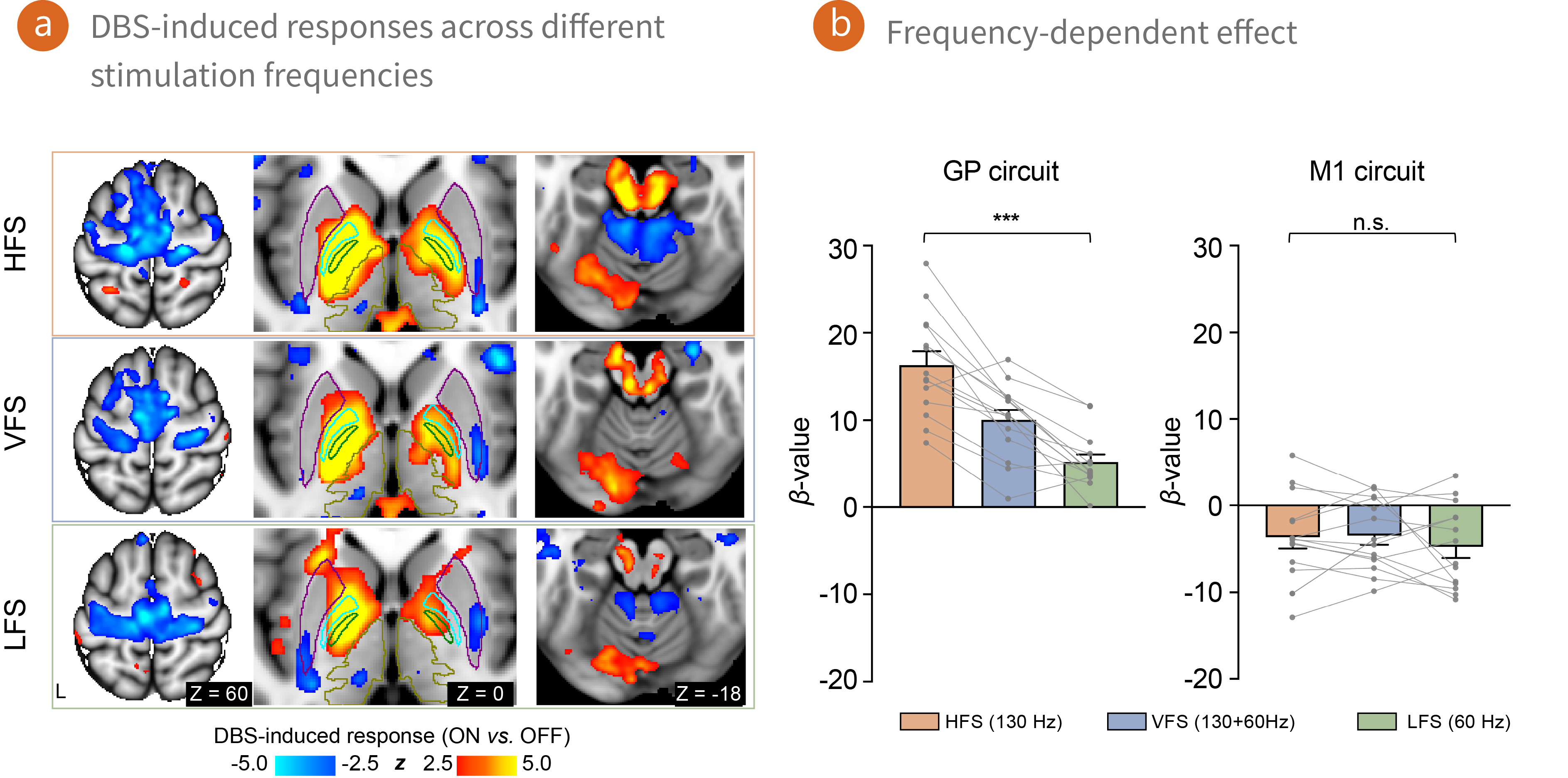
pBFS Application: Functional circuits responding to deep brain stimulation revealed by precision imaging of individual human brains
Deep brain stimulation (DBS) is an effective therapy for treatment-resistant Parkinson’s disease (PD), but its mechanisms and effects on individual brain networks remain poorly understood, underscoring the need for personalized strategies.
Leveraging recent advances in 3T MRI-compatible DBS and precision imaging, we collected a densely-sampled, multi-modal dataset from 14 PD patients undergoing DBS, including 11.7 hours of fMRI across seven stimulation conditions, 2.2 hours of structural MRI, 1.3 hours of diffusion-weighted MRI, and comprehensive neurological assessments over five timepoints spanning one year. Analysis revealed that DBS evoked differential responses in two distinct neurocircuits: the primary motor circuit and the globus pallidus circuit. These findings highlight circuit-specific mechanisms of DBS, providing a foundation for precision-guided neuromodulation and advancing understanding of PD pathophysiology at the network level.
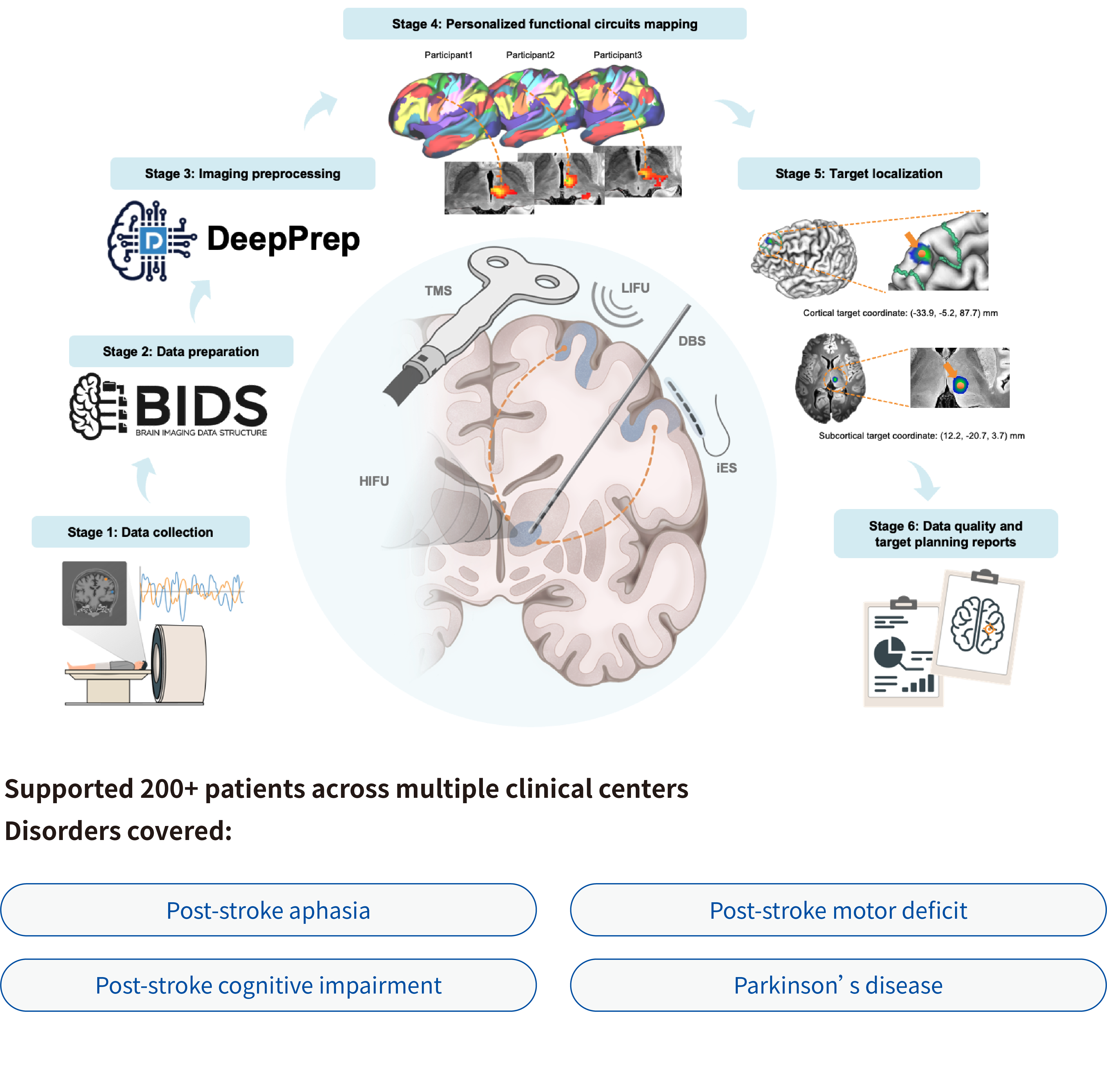
pBFS Protocol: A generic, rapid, and robust platform for personalized functional circuit-guided neuromodulation targeting
Neuromodulation techniques—such as transcranial magnetic stimulation (TMS), focused ultrasound stimulation (FUS), deep brain stimulation (DBS), and intracranial electrical stimulation (iES)—target specific brain functional circuits to treat various brain disorders. Resting-state fMRI (rsfMRI) is increasingly used for individual target localization, but clinical translation is hindered by slow processing, limited robustness, and lack of automated targeting solutions.
We present UNITE(Unified platform for Neuromodulation with Individualized Target Estimation), a rapid, robust, and automated platform for personalized functional circuit-guided neuromodulation. Powered by DeepPrep, UNITE enables precise circuit mapping and automated target localization across multiple modalities. It generates quality reports, exports coordinates compatible with neuronavigation systems, and completes the full workflow within 30 minutes on a standard GPU workstation, advancing scalable, clinically applicable personalized neuromodulation.
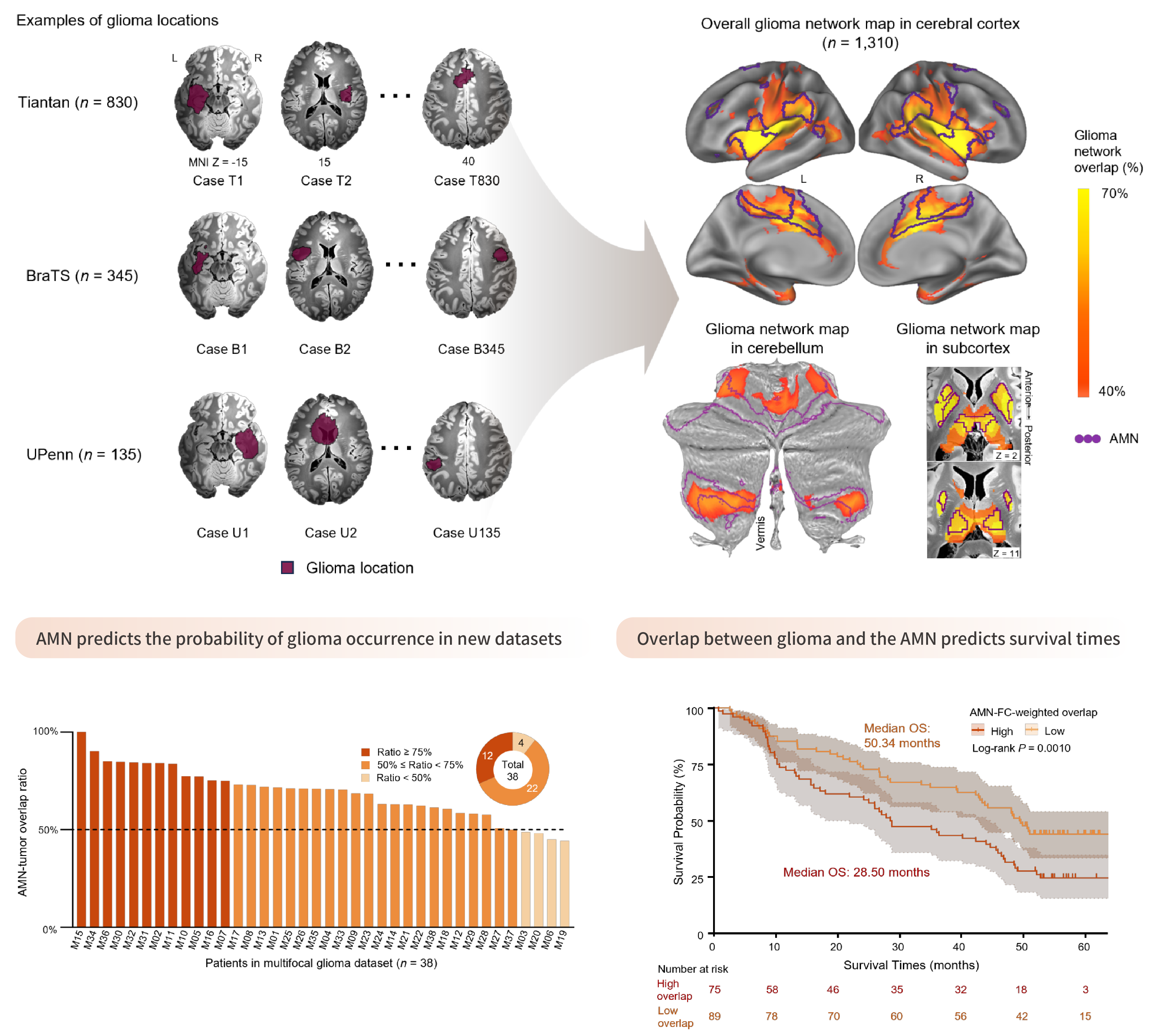
pBFS Mapping: Gliomas preferentially develop within the action-mode network
Gliomas tend to arise in specific brain regions and may integrate into functional circuits, suggesting they could be regulated by brain activity. However, it remains unclear whether glioma growth is related to system-level brain networks.
By analyzing neuroimaging data from three cohorts including 1,310 patients with cerebral gliomas, we identified and replicated a functionally connected glioma network, which overlaps with the action-mode network (AMN), somatomotor network, and action-related subcortical regions. AMN connectivity predicted glioma occurrence in two independent datasets, with over 89% of tumors showing at least 50% overlap with the AMN. Moreover, greater AMN-tumor overlap correlated with poorer survival (n=285). These findings indicate that gliomas preferentially arise within action-related brain circuits, highlighting the central role of the AMN in glioma pathophysiology and potential as a network-level target for pBFS therapy.